Quality of life and strength, but not cytokines, are associated with DAS-28 in active and inactive rheumatoid arthritis
Main Article Content
Abstract
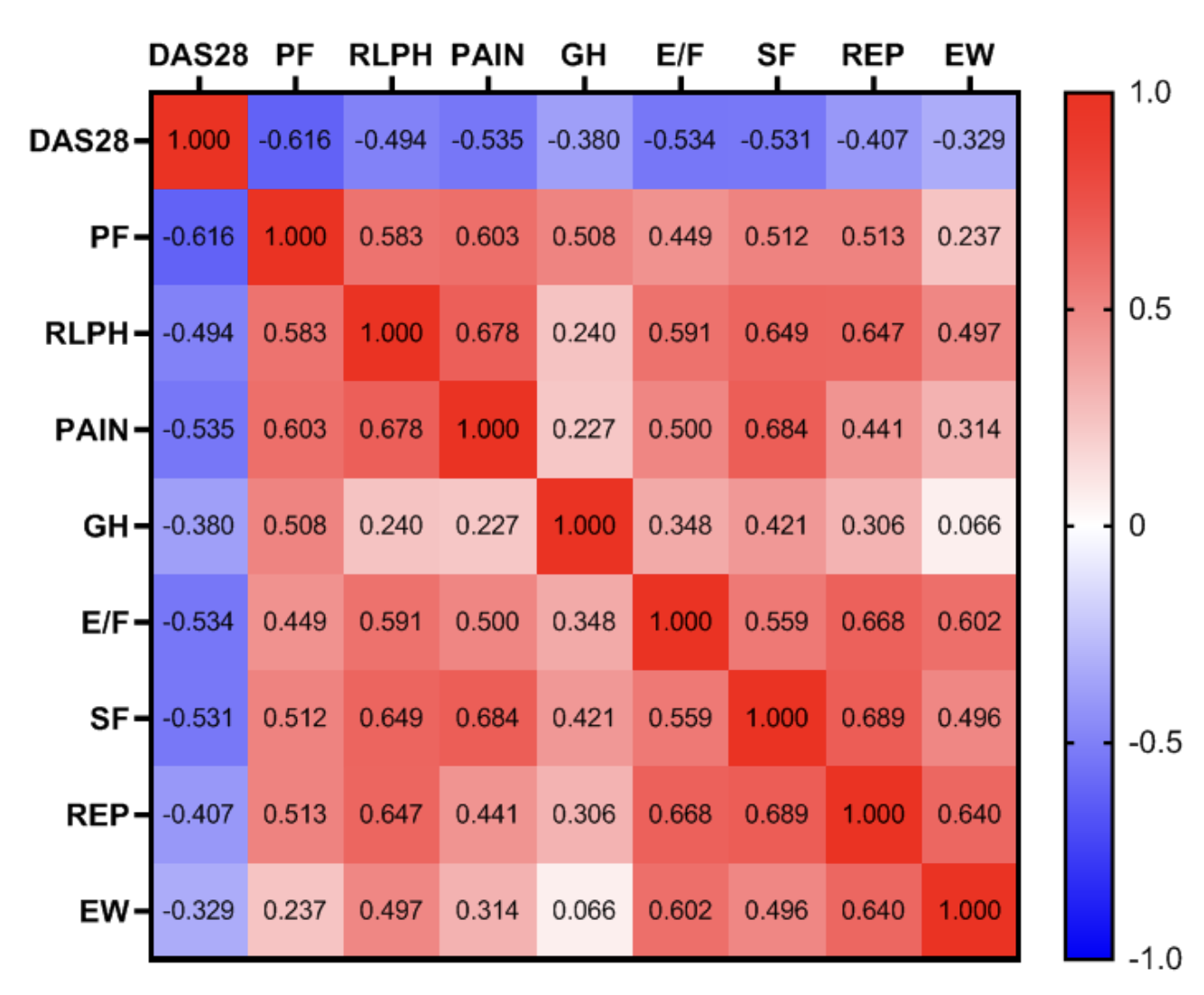
Objective: Evaluate and correlate data between relevant cytokines, disease progression, and handgrip and quality of life among RA patients at different stages of disease progression. Method: Thirty-three RA patients were recruited for analysis, using comparisons and correlations, between levels of circulating cytokines (IFN-γ, TNF-α, IL-4, IL-6, IL-10, IL-17, IL-1β, and TNF receptors I and II), activity of the disease (evaluated using the DAS-28), handgrip (Hydraulic dynamometer), and quality of life (SF-36). Result: RA patients in different disease stages showed increases of IL-6 and IL-10 compared control group. Positive correlation between IL-6 with TNF-α, and IL-4 with IL-10 was found. Handgrip strength and quality of life were not related to cytokine levels. However, remission patients had better strength and quality of life indices compared to the active patients. In addition, handgrip of the non-dominant side, physical functions, role limitations physical health, pain, energy/fatigue and social functions have a negative correlation with the DAS28-PCR. Conclusion: High levels of IL-6 and IL-10 were observed in the chronic RA patients, but the values did not show correlation with disease activity, handgrip strength and quality of life. Disease activity show correlation with handgrip strength and quality of life. Furthermore, remission patients had better strength and quality of life indices compared to the active patients.
Article Details
Authors maintain copyright and grant the HSJ the right to first publication. From 2024, the publications wiil be licensed under Attribution 4.0 International 
 , allowing their sharing, recognizing the authorship and initial publication in this journal.
, allowing their sharing, recognizing the authorship and initial publication in this journal.
Authors are authorized to assume additional contracts separately for the non-exclusive distribution of the version of the work published in this journal (e.g., publishing in an institutional repository or as a book chapter), with acknowledgment of authorship and initial publication in this journal.
Authors are encouraged to publish and distribute their work online (e.g., in institutional repositories or on their personal page) at any point after the editorial process.
Also, the AUTHOR is informed and consents that the HSJ can incorporate his article into existing or future scientific databases and indexers, under the conditions defined by the latter at all times, which will involve, at least, the possibility that the holders of these databases can perform the following actions on the article.
References
Derksen VFAM, Ajeganova S, Trouw LA, van der Helm-van Mil AHM, Hafström I, Huizinga TWJ, et al. Rheumatoid arthritis phenotype at presentation differs depending on the number of autoantibodies present. Ann Rheum Dis 2017;76(4):716- 20. http://doi.org/10.1136/annrheumdis-2016-209794. PMid:28283528. DOI: https://doi.org/10.1136/annrheumdis-2016-209794
Gavrilă BI, Ciofu C, Stoica V. Biomarkers in rheumatoid arthritis, what is new? J Med Life 2016;9(2):144-8.
Ma MHY, Defranoux N, Li W, Sasso EH, Ibrahim F, Scott DL, et al. A multi- biomarker disease activity score can predict sustained remission in rheumatoid arthritis. Arthritis Res Ther 2020;22(1):158. http://doi.org/10.1186/s13075-020-02240-w. PMid:32580789. DOI: https://doi.org/10.1186/s13075-020-02240-w
Hambardzumyan K, Saevarsdottir S, Forslind K, Petersson IF, Wallman JK, Ernestam S, et al. A multi‐biomarker disease activity score and the choice of second‐line therapy in early rheumatoid arthritis after methotrexate failure. Arthritis Rheumatol 2017;69(5):953-63. http://doi.org/10.1002/art.40019. PMid:27992691. DOI: https://doi.org/10.1002/art.40019
Noack M, Miossec P. Selected cytokine pathways in rheumatoid arthritis. Semin Immunopathol 2017;39(4):365-83. http://doi. org/10.1007/s00281-017-0619-z. PMid:28213794. DOI: https://doi.org/10.1007/s00281-017-0619-z
Milman N, Karsh J, Booth RA. Correlation of a multi-cytokine panel with clinical disease activity in patients with rheumatoid arthritis. Clin Biochem 2010;43(16-17):1309-14. http://doi. org/10.1016/j.clinbiochem.2010.07.012. PMid:20655893. DOI: https://doi.org/10.1016/j.clinbiochem.2010.07.012
Reis MM, Paula MMA. Medida da força de preensão manual-validade e confiabilidade do dinamômetro saehan. Fisioter Pesqui. 2011;18(2):176-81. http://doi.org/10.1590/S1809- 29502011000200013.
Ramírez-Vélez R, Sáez De Asteasu ML, Martínez-Velilla N, Zambom-Ferraresi F, García-Hermoso A, Recarey AE, et al. Circulating cytokines and lower body muscle performance in older adults at hospital admission. J Nutr Health Aging 2020;24(10):1131-9. http://doi.org/10.1007/s12603-020-1480-7. PMid:33244573. DOI: https://doi.org/10.1007/s12603-020-1480-7
Yen DH-T. Handgrip strength, tumor necrosis factor-α, interlukin-6, and visfatin levels in oldest elderly patients with cognitive impairment. Exp Gerontol 2020;142:111138. http:// doi.org/10.1016/j.exger.2020.111138. PMid:33122129. DOI: https://doi.org/10.1016/j.exger.2020.111138
Beenakker KGM, Ling CH, Meskers CGM, de Craen AJM, Stijnen T, Westendorp RGJ, et al. Patterns of muscle strength loss with age in the general population and patients with a chronic inflammatory state. Ageing Res Rev 2010;9(4):431-6. http:// doi.org/10.1016/j.arr.2010.05.005. PMid:20553969. DOI: https://doi.org/10.1016/j.arr.2010.05.005
Medeiros MMC, Oliveira BMGB, Cerqueira JVM, Quixadá RTS, Oliveira ÍMAX. Correlação dos índices de atividade da artrite reumatoide (Disease Activity Score 28 medidos com VHS, PCR, Simplified Disease Activity Index e Clinical Disease Activity Index) e concordância dos estados de atividade da doença com vários pontos de corte numa população do nordeste brasileiro. Rev Bras Reumatol 2015;55(6):477-84. http://doi.org/10.1016/j. rbr.2014.12.005. DOI: https://doi.org/10.1016/j.rbr.2014.12.005
American Society Hand Therapy. The hand: examination and diagnosis. 3rd ed. New York: Churchill Livingstone; 1983.
Reis MM, Arantes PMM. Medida da força de preensão manual-validade e confiabilidade do dinamômetro saehan. Fisioter Pesqui 2011;18(2):176-81. http://doi.org/10.1590/S1809- 29502011000200013. DOI: https://doi.org/10.1590/S1809-29502011000200013
Cicconeli RM, Ferraz MB, Santos W, Meinão I, Quaresma MR. Brazilian-Portuguese version of the SF-36. A reliable and valid quality of life outcome measure. Rev Bras Reumatol 1999;39:143-50.
Senolt L. Emerging therapies in rheumatoid arthritis: focus on monoclonal antibodies. F1000Res 2019;8:F1000 Faculty Rev- 1549. http://doi.org/10.12688/f1000research.18688.1. PMid: 31508202. DOI: https://doi.org/10.12688/f1000research.18688.1
Gerli R, Bistoni IO, Russano A, Fiorucci IS, Borgato L, Cesarott MEF, et al. In vivo activated T cells in rheumatoid synovitis. Analysis of Th1- and Th2-type cytokine production at clonal level in different stages of disease. Clin Exp Immunol 2002;129(3):549-55. http://doi.org/10.1046/j.1365- 2249.2002.01913.x. PMid:12197898. DOI: https://doi.org/10.1046/j.1365-2249.2002.01913.x
Barnes PJ. Anti-inflammatory actions of glucocorticoids: molecular mechanisms. Clin Sci (Lond) 1998;94(6):557-72. http://doi.org/10.1042/cs0940557. PMid:9854452. DOI: https://doi.org/10.1042/cs0940557
Relić B, Guicheux J, Mezin F, Lubberts E, Togninalli D, Garcia I, et al. IL-4 and IL-13, But Not IL-10, protect human synoviocytes from apoptosis. J Immunol 2001;166(4):2775-82. http://doi. org/10.4049/jimmunol.166.4.2775. PMid:11160344. DOI: https://doi.org/10.4049/jimmunol.166.4.2775
Morita Y, Yamamura M, Kawashima M, Aita T, Harada S, Okamoto H, et al. Differential in vitro effects of IL-4, IL-10, and IL-13 on proinflammatory cytokine production and fibroblast proliferation in rheumatoid synovium. Rheumatol Int 2001;20(2):49-54. http://doi.org/10.1007/s002960000074. PMid:11269532. DOI: https://doi.org/10.1007/s002960000074
Marinou I, Healy J, Mewar D, Moore DJ, Dickson MC, Binks MH, et al. Association of interleukin-6 and interleukin-10 genotypes with radiographic damage in rheumatoid arthritis is dependent on autoantibody status. Arthritis Rheum 2007;56(8):2549-56. http://doi.org/10.1002/art.22814. DOI: https://doi.org/10.1002/art.22814
Byun MK, Cho EN, Chang J, Ahn CM, Kim HJ. Sarcopenia correlates with systemic inflammation in COPD. Int J Chron Obstruct Pulmon Dis 2017;12:669-75. http://doi.org/10.2147/ COPD.S130790. PMid:28255238. DOI: https://doi.org/10.2147/COPD.S130790
Torii M, Hashimoto M, Hanai A, Fujii T, Furu M, Ito H, et al. Prevalence and factors associated with sarcopenia in patients with rheumatoid arthritis. Mod Rheumatol 2019;29(4):589-95. http://doi.org/10.1080/14397595.2018.1510565. DOI: https://doi.org/10.1080/14397595.2018.1510565
Espinoza F, Le Blay P, Coulon D, Lieu S, Munro J, Jorgensen C, et al. Handgrip strength measured by a dynamometer connected to a smartphone: a new applied health technology solution for the self-assessment of rheumatoid arthritis disease activity. Rheumatology (Oxford) 2016;55(5):897-901. http:// doi.org/10.1093/rheumatology/kew006. PMid:26867731. DOI: https://doi.org/10.1093/rheumatology/kew006
Caporrino FA, Faloppa F, Santos JBG, Réssio C, Soares FHC, Nakachima LR, et al. Estudo populacional da força de preensäo palmar com dinamômetro Jamar. Rev Bras Ortop 1998;33(2):150-4.
Mitsionis G, Pakos EE, Stafilas KS, Paschos N, Papakostas T, Beris AE. Normative data on hand grip strength in a Greek adult population. Int Orthop 2009;33(3):713-7. http://doi. org/10.1007/s00264-008-0551-x. PMid:18414855. DOI: https://doi.org/10.1007/s00264-008-0551-x
Adedoyin RA, Ogundapo FA, Mbada CE, Adekanla BA, Johnson OE, Onigbinde TA, et al. Reference values for handgrip strength among healthy adults in Nigeria. Hong Kong Physiother J 2009;27(1):21-9. http://doi.org/10.1016/S1013-7025(10)70005-1. DOI: https://doi.org/10.1016/S1013-7025(10)70005-1
Wang Y-C, Bohannon RW, Li X, Sindhu B, Kapellusch J. Hand-grip strength: normative reference values and equations for individuals 18 to 85 years of age residing in the United States. J Orthop Sports Phys Ther 2018;48(9):685-93. http://doi. org/10.2519/jospt.2018.7851. PMid:29792107. DOI: https://doi.org/10.2519/jospt.2018.7851
Azevedo AFB, de Petribú KCL, Lima MN, Silva AS, Rocha JA Fo, Mariano MHQA, et al. Quality of life of patients with rheumatoid arthritis under biological therapy. Rev Assoc Med Bras 2015;61(2):126-31. http://doi.org/10.1590/1806- 9282.61.02.126. PMid:26107361. DOI: https://doi.org/10.1590/1806-9282.61.02.126
Azevedo LF, Costa-Pereira A, Mendonça L, Dias CC, Castro- Lopes JM. Epidemiology of chronic pain: a population-based nationwide study on its prevalence, characteristics and associated disability in Portugal. J Pain 2012;13(8):773-83. http://doi.org/10.1016/j.jpain.2012.05.012. PMid:22858343. DOI: https://doi.org/10.1016/j.jpain.2012.05.012
Campos APR, Silva CM, de Castro SS, Graminha CV. Depressão e qualidade de vida em indivíduos com artrite reumatoide e indivíduos com saúde estável: um estudo comparativo. Fisioter Pesqui 2013;20(4):401-7. http://doi.org/10.1590/ S1809-29502013000400016. DOI: https://doi.org/10.1590/S1809-29502013000400016
Salaffi F, Carotti M, Gasparini S, Intorcia M, Grassi W. The health-related quality of life in rheumatoid arthritis, ankylosing spondylitis, and psoriatic arthritis: a comparison with a selected sample of healthy people. Health Qual Life Outcomes 2009;7(1):25. http://doi.org/10.1186/1477-7525- 7-25. PMid:19296831. DOI: https://doi.org/10.1186/1477-7525-7-25